Bacteria, viruses and other germs sometimes set off the immune system to overreact, producing a severe condition called sepsis. Sepsis is so dangerous that it is the leading cause of death of children across the world, killing a million kids every year, mostly in developing countries. Probiotic bacteria might be able to prevent sepsis and infections, but no large research studies have been done to find out whether that actually works. Until now.
A research study done on 4556 infants in India found that synbiotics — combinations of probiotics with a prebiotic, which feeds the probiotics and promote their growth — given to newborns cut the occurrence of sepsis by 40%.
The synbiotics were given to the newborns for seven days within the first two weeks of their life. After 60 days of follow up, sepsis was significantly reduced in babies given synbiotics, compared to babies not given them. The cost of the treatment was only about $1 a day per baby.
The study, led by Pinaki Panigrahi, from the University of Nebraska Medical Center, and his colleagues was published August 16 in Nature.
The Devastation That Is Pediatric Sepsis
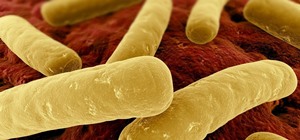
Sepsis in a newborn is most often the result of bacteria in their blood. E. coli, Listeria, and some strains of streptococcus, especially Group B streptococcus, have been identified as major causes of neonatal sepsis. Screening of women in the US for Group B strep has cut neonatal sepsis rates in this country.
Mothers infect their newborns before or during delivery, so newborn sepsis appears within the first day of birth. Changes in body temperature, breathing problems, diarrhea, low blood sugar, reduced movement or sucking, seizures, fast or slow heart rate, swollen belly, vomiting, and jaundice — yellow skin or whites of the eyes — are all signs of newborn sepsis.
Low birthweight and premature birth increase the risk of an infant developing sepsis. About a fourth of all babies in the US born weighing less than 3.3 pounds have an infection that leads to sepsis. However, in sub-Saharan Africa, South Asia, and Latin America 3.5 million babies develop a severe infection every year and 8–80% of them die from those infections, many of which had led to sepsis — even with antibiotic treatment.
This is a tragic situation compounded by economic conditions in developing countries that make expensive treatments out of the question. Panigrahi and his colleagues may have come up with a way to prevent most of those infections from occurring, and it costs just a dollar a day.
Huge Health Return on $1 Investment
To boost the immune system of newborns, Pangrahi and his team designed a study to give the infants bacteria that would colonize — take up residence and grow in — their gut and hopefully prevent infections.
The new research study, conducted on 4556 infants in rural India, tested the effect of a synbiotic on newborn sepsis and death rates. Researchers gave the infants a synbiotic containing Lactobacillus plantarum bacteria and a sugar called fructooligosaccharide, which acted as food for the bacteria. An equal number of infants (2278) received a placebo that contained no synbiotic. The synbiotics and placebos were prepared as powders and supplied in a capsule that was dissolved in a sterile sugar and salt solution and given to the babies for seven days in the first two weeks of their life. Community volunteers checked on the babies for 60 days for signs of severe infection or sepsis. If they found signs or symptoms of infection, they referred the infants to hospitals that were involved in the study.
After 60 days, 5.4% of the infants who received synbiotics had suffered from sepsis, while 9% of those who received placebo had. There were 10 deaths total, six in those getting the treatment and four in the placebo group. One child from each group died of hypothermia. Staphylococcus aureus was isolated from one baby who died in each group and probably contributed to the deaths of those two infants. Children receiving placebo, some of whom died, were more frequently infected Staphylococcus aureus, E. coli, Streptococcus viridans or Klebsiella pneumoniae than children who were treated with synbiotics.
Before synbiotics could be recommended for all newborns, the study authors suggested that they should be tested in premature babies, newborns with severe medical conditions at birth, and other babies at high risk for sepsis. The probiotic and prebiotic seemed to work on the infants in this study, but the authors noted that other populations of infants might need different formulations. Other hurdles to a large scale program are maintaining cold shipment and storage of the synbiotic, and getting the preparation to all newborns in all areas.
Given the promising results and affordable price of this preventative treatment, follow-up studies and development of ways to meet the logistical challenges should be given priority to move this treatment from the research realm to all newborns.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.




















Be the First to Comment
Share Your Thoughts