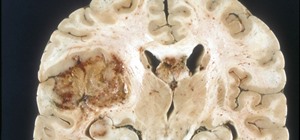
Prion diseases are a group of infectious brain diseases that causes extensive tissue damage, resulting in sponge-like spaces in brain tissue. Prions include Creutzfeldt-Jakob disease in humans, bovine spongiform encephalopathy (often called mad cow disease), and chronic wasting disease in hoofed ruminant mammals.
Creutzfeldt-Jakob disease kills about 300 people in the US every year, and about 1 million worldwide. These rare, degenerative prion diseases of the brain are always fatal. Long periods of time elapses between infection and development of symptoms—sometimes as long as three decades. There has been no way to tell if a patient has a prion disease and the disease is usually diagnosed at autopsy.
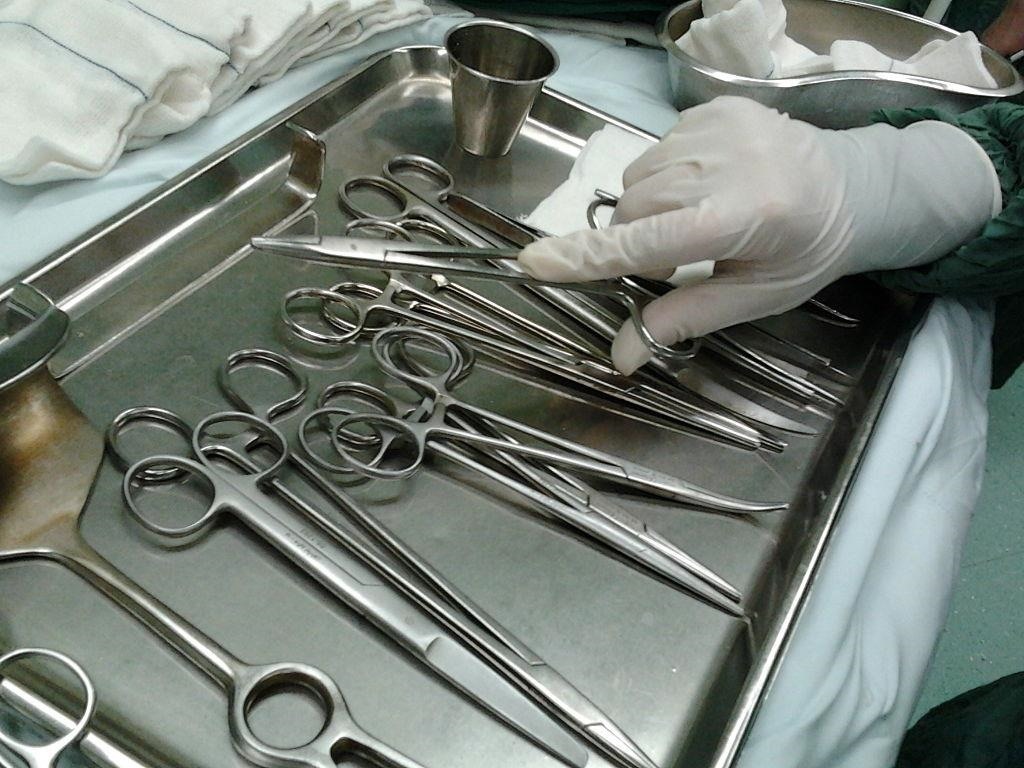
This is a huge problem because, even though about 85% of cases appear randomly, seemingly without reason, the infectious prion proteins can be transmitted via blood transfusion, blood products, organ or tissue grafts, and contaminated medical instruments (and the proteins are incredibly hardy—they aren't destroyed by normal disinfection routines).
But now we can finally test for these infections. Researcher Byron Caughey, a biochemist at Rocky Mountain Laboratories (a part of the NIH Intramural Research Program), and his colleagues, have developed a rapid and sensitive test that can detect Creutzfeldt-Jakob disease using cerebrospinal fluid or nasal brushing samples (scrapings taken at a spot where the nose is separated from the brain by a small tissue divider).
Results of testing the new assay were recently published in the Annals of Neurology.
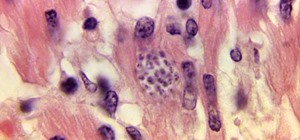
Prions themselves are a protein found throughout the body, but are greatest in number in the brain. Researchers aren't completely clear on what normal prion proteins do, but studies have suggested they play a role in nerve repair, long-term memory, and/or stem cell renewal.
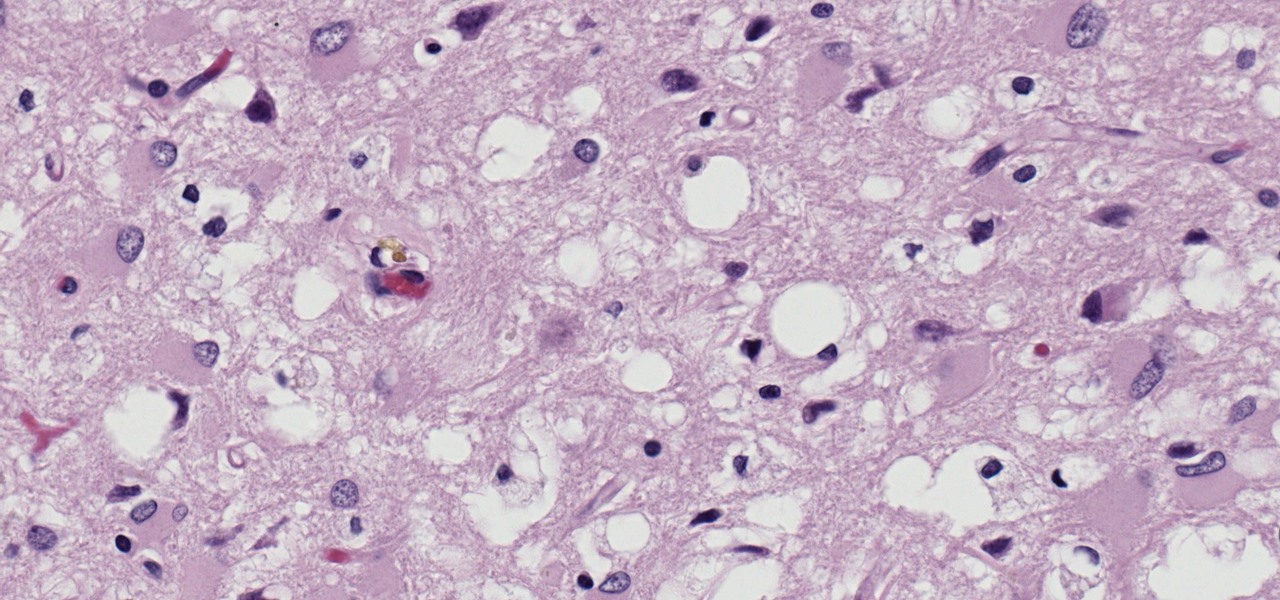
But what's really fascinating about these proteins is that if they're misfolded—bent into a different shape inside the cell—they can interact with normally folded prions and misfold them, passing on the structural change. Both of those bent prions can move on and misfold more prions in the cell, the body, and other bodies, making the protein "infectious" like a virus. Abnormally folded proteins clump up, kill brain cells, and lead to brain damage and dementia.
Somehow, the way the prion is misfolded in the infectious particle makes it immune to almost every kind of disinfection; It can withstand heat, pressure, boiling, ultraviolet light, and chemicals used for disinfecting, like formaldehyde, and still be infectious.
That means it can be passed on without anyone knowing, like in the Middlesbrough General Hospital scare in the UK. In 2002, 24 people worried they might have contracted Creutzfeldt-Jakob disease after finding that surgical instruments used for their surgery were previously used on a woman later found to have the prion disease.
Infection with prion disease through use of contaminated medical products or equipment isn't common, but it does happen, and when it does, it's usually because of one undiagnosed patient. And because the incubation period is so long, that person could have crossed paths with the patient (or their prions) decades before showing symptoms.
Five patients in the UK had developed Creutzfeldt-Jakob disease after surgical procedures on their brains were conducted using instruments previously used for neurosurgical operations on a patient later discovered to have Creutzfeldt-Jakob disease. Another two patients were infected with Creutzfeldt-Jakob disease as a result of corneal surgery.
Between 1958 and 1985, human growth hormone was produced by extracting it from large numbers of pituitary glands, then given to thousands of children across the globe. These small glands at the base of the brain were obtained from bodies after death, and some of these gland donors were later found to have prion disease that infected the growth hormone recipients.
After 1985, a synthetic form of growth hormone became available. However, every year, two to six young adults in the UK who were treated with growth hormone as children before 1985 are diagnosed with Creutzfeldt-Jakob disease.
Once the symptoms of prion disease—rapidly developing dementia, difficulty walking, hallucinations, muscle stiffness, confusion, fatigue, and difficulty speaking—develop, the infection may already have been present for decades and the patient deteriorates rapidly, dying within eight to 18 months.
Breakthrough for Prion Disease Diagnosis—& Probably Others
The new test uses patient samples—either cerebrospinal fluid or nasal brushings—that are mixed with normal brain tissue in a tissue culture plate, then shaken. If prions are present, shaking the reaction mixture speeds up the misfolding and clumping of brain proteins. After 90 hours, the tissue is examined to see if the normal brain proteins misfolded. Misfolded proteins means prion disease is present. The researchers named the test, RT-QuIC, for "real-time quaking-induced conversion."
They tested samples from 2,141 patients with the new and improved test. The test was highly sensitive, correctly identifying nearly 100% of samples from patients known to have Creutzfeldt-Jakob disease. The test correctly reported 92-95% of the samples without the disease as negative for the disease.
The new test is a breakthrough for use in diagnosing patients with prion diseases, but neurodegenerative disease researchers aren't stopping there. Other brain diseases that work similarly—by created clumps of proteins in in brain—could be tested for in similar ways.
Dr. Alison Green, a biochemist at the University of Edinburgh in the UK, is currently adapting the test to detect two other neurodegenerative diseases—Parkinson's disease and Lewy body dementia.
She already has preliminary results that indicated the modified test correctly identified 19 out of 20 samples from patients with Parkinson's disease. Dr. Green has applied for funding to develop similar testing for Alzheimer's disease.
She hopes to develop a wide variety of RT-QuIC assays, so that patients coming in with dementia of unknown origin can get a diagnosis or rule out some causes.
So what does this mean for patients?
Although it's not curable yet, unlike prion diseases, the symptoms of Parkinson's are treatable. Earlier diagnoses and earlier treatment may change the course of the patients' disease. It may do the same for Alzheimer's disease.
Does the new prion test help treat the patient? Not directly, but it does help eliminate other diagnoses, may prevent other inadvertent infections, and can answer an important question for the family.
As the husband of a wife diagnosed with Creutzfeldt-Jakob disease said, "We really, truly wanted to know if there was something that we could do for her." After getting the definitive diagnosis of prion disease, the family knew that supporting her through the end of her life was what they could do.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.



















Be the First to Comment
Share Your Thoughts