Over 1.2 million people in the US are infected with human immunodeficiency virus (HIV)—and one out of eight of them don't know it. Even after decades of intense research into the virus, there's still no cure for it. One of the big problems is that the virus hides out in certain cells of the body, resisting treatments that kill it.
A new study suggests that HIV can infect a type of white blood cell in the brain through a back door and remain hidden there. Finding and activating these repositories can help doctors flush out the virus and kill it for good.
What We Already Knew About HIV
HIV affects people of every gender and age across the world. In the US, young African American gay and bisexual men are most affected. In 2015, 39,513 new cases were diagnosed, and that represents a 19% decrease since 2005.
The HIV epidemic is far from over, but thanks to antiretroviral treatment, it can usually be controlled. Controlled—not cured.
The HIV virus attacks the very cells of the immune system that exist to protect us from pathogens—the CD4(+) T lymphocytes. Once infected, the virus uses the host cell to replicate itself and kills the cell in the process. If untreated, enough of these immune cells die that a person is left very susceptible to pathogens, develops an infection that can't be cured, and dies.
But in addition to infecting and killing white blood cells, scientists know that HIV can hide in what are called latent reservoirs of virus. When this happens, the level of HIV found in the blood are very low—so low they're undetectable—but if treatment is stopped, the virus can com raging back.
The virus stays hidden inside the cells of the central nervous system, the male urogenital tract, and in CD4(+) T cells by storing a copy of its genetic code in the cell's genome. At the drop of a hat, this viral code can reactivate.
In some people, HIV hides out and replicates in macrophages in the brain. Macrophages are much harder for HIV to infect than CD4+ T cells because they make a special antiviral protein. But in a small percentage of macrophages, HIV finds a way in and hides out.
Figuring out how this invasion happens could be the last piece in HIV-cure puzzle.
New findings by a group of researchers from the United Kingdom, South Africa, and Atlanta, Georgia, to be published in EMBO Journal, revealed a glitch in the macrophage's protective armor that may be exploited as a way to cure the body of latent HIV infections.
The Lesser Player May Have the Most to Teach Us
White blood cells like macrophages, dendritic cells, and CD4(+) T cells spend their time surveilling the body's mucosal surfaces, and are the first immune cells that run into HIV when it infects someone.
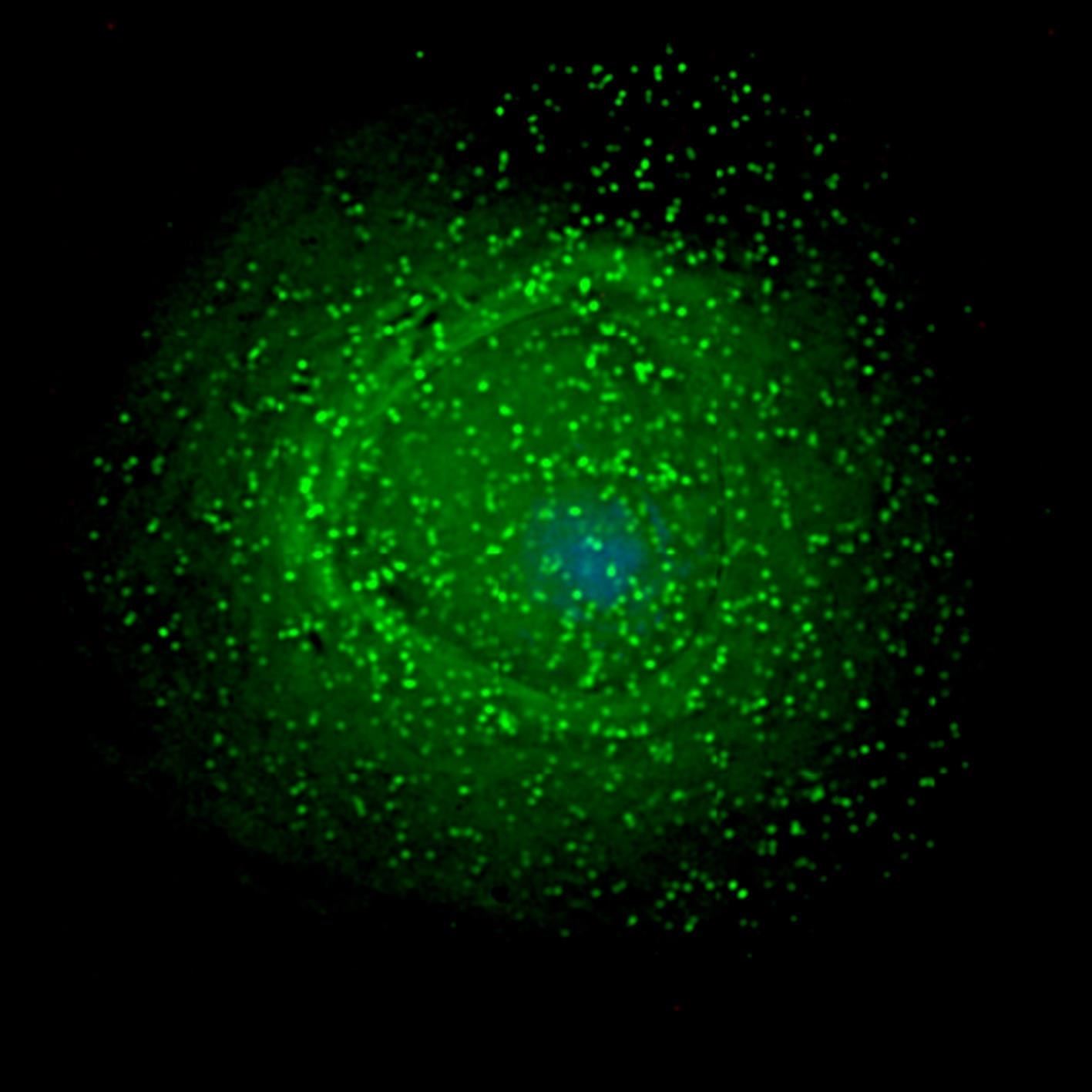
All three types of white blood cells can become infected with HIV, but only a small number of dendritic cells and macrophages get a productive infection—one in which the virus is replicated and spewed out into the blood.
One reason macrophages are harder to infect with HIV than CD4(+) cells is because they make their own antiviral protein, called SAMHD1. This protein, also made by white blood cells like CD4(+) lymphocytes, prevents HIV from replicating, but is switched off when the cells multiply. Since macrophages don't multiply, researchers believed they never switched off their SAMHD1, so were always protected from infection.
At least, that's what they thought until the discovery by the research team, led by Dr. Petra Mlcochova of the Division of Infection and Immunity at University College London.
"We found there's a window of opportunity when SAMHD1 is disabled as part of a regularly-occurring process in macrophages," senior author Ravindra Gupta, also from the Division of Infection and Immunity at UCL, said in a press release.
Without the antiviral protection of SAMHD1, HIV infection and replication can proceed in the cells during this small window of time—which takes up about 20% of its lifecycle.
Once the team showed that SAMHD1 was disabled in macrophages occasionally, they showed they could be infected with HIV during those times.
Our findings could help explain why some people undergoing anti-retroviral therapy for HIV continue to have HIV replication in the brain, as the infected cells in the brain are typically macrophages. While this is a barrier to achieving control of HIV in just a minority of patients, it may more importantly be a barrier to a cure.
The cellular machinery that keeps the HIV virus lying latent in cells is controlled by a group of enzymes called histone deacetylases (HDAC). HDAC inhibitors have been shown to reactivate latent HIV infections by deactivating the HCAC molecules.
Reactivation of HIV via the use of HDAC inhibitors would, theoretically, let these hidden viruses out, depleting the latent pool, but it also makes the newly-produced virus available to infect other cells. If SAMHD1 and other HIV drugs could then be used to protect cells from infection, the HIV-fighting combo of SAMHD1 and HDAC inhibitors may deliver the fatal blow to the infection, a potential combo for a cure.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.



















Be the First to Comment
Share Your Thoughts