Despite legends to the contrary, it appears that the saliva of a Komodo dragon is not teeming with pathogenic bacteria that kills their prey. Its reputation to survive while colonized with lots of horrible disease-causing bacteria, true or untrue, has made it the subject of research in pursuit of natural antimicrobial agents and led scientists to some remarkable findings.
According to two new research studies led by biochemists Barney Bishop and Monique van Hoek from the College of Science at George Mason University, their blood contains antimicrobial proteins that help them resist deadly infections and may be of potential use to us, too.
Compounds in Dragon Blood Showed Antibacterial Activity
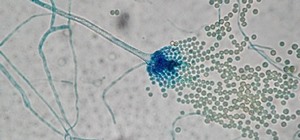
In one study by the team, published in the Journal of Proteome Research, they used a type of bioprospecting — the search for medicinal drugs and other compounds from natural sources — to look for antibacterial compounds in the blood of Komodo dragons.
They were looking for unique compounds called cationic antimicrobial peptides — positively charged pieces of protein. Most animals produce these peptides as part of their immune system, but the researchers hoped the Komodo dragon might be making some unusual ones.
So they incubated Komodo dragon blood with negatively charged particles to attract and pull out the positively charged peptides. They found and analyzed 48 cationic peptides, 47 of which were from proteins known to have antimicrobial properties.
In the lab, the study team tested eight of the compounds against Pseudomonas aeruginosa and Staphylococcus aureus bacteria, which commonly infect skin wounds. Seven of them showed antibacterial activity against both bacteria. The eighth only showed significant potency against P. aeruginosa.
Synthetic Version of Dragon Blood Compound Healed Wounds
Another recent study by the duo expanded on their previous finding and was published online in the journal Biofilms and Microbiomes. The scientists chose one peptide they identified earlier, called VK25, and developed a synthetic version with a structure designed to increase its effectiveness, which they called DRGN-1.
Next, they tested the antimicrobial power of the two peptides against the same two bacteria used in the first study. DRGN-1, the synthetic, enhanced version of the peptide, showed a greater antimicrobial and anti-biofilm activity — disruption of networks bacteria form to communicate and survive — than the original, VK25.
When researchers placed a biofilm of these two bacteria on skin wounds in mice, treatment with DRGN-1 reduced the bacterial counts and completely healed the infections after 11 days. When treated with VK25, the wounds did not heal in 11 days.
The results — that DRGN-1 kills bacteria, disrupts biofilms, and helps wounds heal — are encouraging. "These features suggest that DRGN-1 might be developed as a topical agent for infected wound treatment," the researchers wrote in their paper.
Bioprospecting the Komodo dragon's blood may bring a mythical animal to the forefront of the very real battle to find new, more effective antimicrobials.
Real-Life Dragons with a Larger Than Life Reputation
Komodo dragons are the world's largest lizards, but most of us will never see them because they live only on five small islands in Indonesia.
Controversy rages over whether the Komodo dragon kills its prey by injecting and infecting it with nasty bacteria in its saliva — and whether it has superpowers to survive living with all those pathogens. The animal's ability to take down animals as large as water buffalo bolster that legend.
A study of the saliva of 16 captive Komodo dragons found 128 types of bacteria, but none of them were disease-causing. The only pathogenic bacteria found in their mouths ended up there from the bacteria-filled watering holes where the Komodo dragons drank.
Another study found Komodo dragons deliver venom by forcefully biting their prey, then injecting venom into the wound in a specialized motion that ensures the venom lands right where they want it — in the wound.
We can't put the legend of Komodo dragons' power to rest yet. The dispute over whether its power lies in venom, bacteria, cunning, or a combination of all those things will remain part of its mystique.
One thing we now know for sure, though, thanks to Barney Bishop and Monica van Hoek, is that their blood may give us a new weapon to use against bacterial infections.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.






















2 Comments
That's fascinating research, I always suspected hat was why komodo dragons had been able to survive for so long and in such isolation,but I never thought that humans would Some day benefit from they're immune system. That's really amazing work by the scientific branches that performed this research,much applauded
It is amazing how creatures like this one even exist, I mean it only proves that all the species are interconnected in a symbiotic way. Unfortunately, in the last few years there's a decline of the komodo dragon populations. Hope it doesn't become completely extinct.
Share Your Thoughts