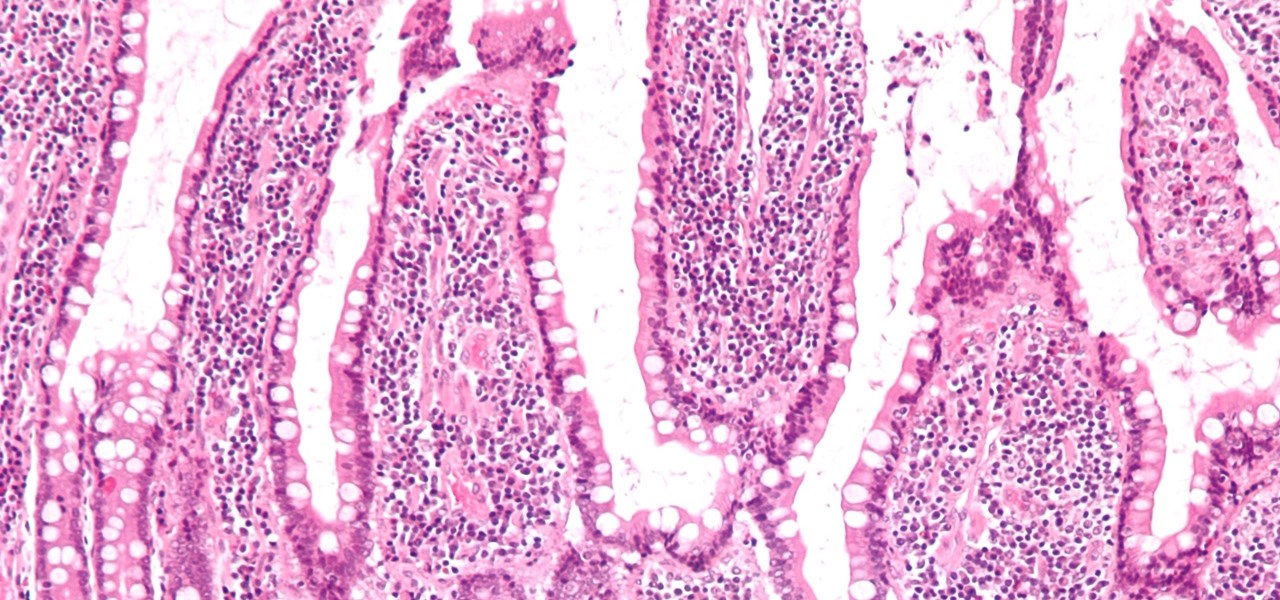
If the all the fingerlike projections in our gut were flattened out, its surface area would be 100 times bigger than our skin's. It's so large that the actions of just a small part of it can impact our health. A new research study has found that enterochromaffin cells in the intestinal lining alert the nervous system to signs of trouble in the gut — trouble that ranges from bacterial products to inflammatory food molecules.
The new research, led by scientists from UC San Francisco, discovered that these enterochromaffin cells have sensors that trigger electrical impulses in nearby nerve fibers when they detect three types of materials: chemicals called fatty acids released from bacteria, hormones that signal stress, and a dietary irritant. It's a direct line from the gut to the brain.
The research findings were published June 22 online in Cell.
"These cells are sensors, like a window looking into the contents of the gut," lead author James Bayrer, from UCSF, said in a press release.
Our 'Second Brain'
Usually, nerve signals that control organ functions come directly from the brain. But, that isn't true for our gut. The nerve signals that control the gut come from a network of nerves within the gut itself, earning it the nickname of the "second brain". The nerve impulses are involved in the gut's normal contractions and digestive activities and operate without input from the brain. A different two-way communication between the brain and gut sends messages back and forth along another network called the brain-gut axis which helps us react to what's going on in our gut.
Fingerlike projections called villi line our intestinal tract, and enterochromaffin cells stud their surface. Nerve fibers run underneath the enterochromaffin cells, sensing the movement and contents of the gut and sending that info on to the brain. These signals can cause contractions of the gut or alert us to hunger, stress, or other distress that may be brewing.
Researchers didn't fully understand exactly how nerve fibers and enterochromaffin cells communicate these messages until this new research.
The team thought that nerve signals starting out in the enterochromaffin cells could interact with both networks to cause, for example, diarrhea-causing gut contractions when we get the credit card bill or, if the signals reach the brain, that sinking feeling in the pit of our stomach.
How Enterochromaffin Cells Sense and Alert
To test how the nerve impulses travel from the enterochromaffin cells, the scientists used mice cells grown in the lab to mimic the organization and types of cells in the intestinal tract. They exposed the organ-like cells to dozens of different types of compounds and found that three types of molecules triggered the cells to produce an electrical impulse. These triggers include: bacterial byproducts called volatile fatty acids; a class of hormones called catecholamines like dopamine, epinephrine, and norepinephrine that can signal stress in the gut; and a dietary irritant called AITC which is responsible for garlic's flavor.
When those molecules activated the enterochromaffin cells, the cells released serotonin into the space adjoining nearby nerve fibers. It was this serotonin release, the team found, that triggered electrical impulses in nerve fibers that moved quickly throughout the gut, then to the brain. In the brain, serotonin is involved in mood and behavior, but in the gut, it can generate contractions and discomfort.
Noting that mice cells could be sensitive to different compounds than humans, the researchers next step is to study enterochromaffin cells in human organ-like cells. However, research has linked all three types of molecules found to activate enterochromaffin cells to irritable bowel syndrome in humans. So the scientists think it's likely they will find the same processes at work in people.
The study authors hope that understanding more about how enterochromaffin cells work in people might explain how they trigger normal reactions that cause contractions that push out unwanted chemicals and microbes — at one end of the intestinal tract or the other — or may overreact to cause disorders like irritable bowel syndrome.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.



















Be the First to Comment
Share Your Thoughts