Joe McKenna died when he was 30 years old. A young married man with his future ahead of him, he was cleaning up the station where he worked as a fireman. Struck by a piece of equipment fallen from a shelf, Joe complained of a sore shoulder. Over the next week, Joe worsened and ended up in the hospital. Chilled, feverish, and delirious, his organs shut down from an infection we'd now call septic shock.
The year was 1938, and antibiotics weren't invented until a few years later. Sudden deaths, like that of McKenna, happened frequently. Families were large in the hopes some would survive.
That was the story that journalist and author Maryn McKenna shared in a 2015 TED Talk the about how her great-uncle passed away.
My great-uncle died in the last days of the pre-antibiotic era. We stand today on the threshold of the post-antibiotic era, in the earliest days of a time when simple infections such as the one Joe had will kill people once again.
Bacteria have developed resistance to antibacterial treatments quickly over the decades, and now many of these infections are once again deadly. Although it is a big topic, the basics are important to grasp in order to understand this fast-developing issue.
A Tiny Discovery
Microorganisms are a big part of our personal and physical world. In the 17th century, citizen-scientist Antonie van Leeuwenhoek pursued his passion for examining the world of the microscopic.
By inventing specialized lenses, Van Leeuwenhoek was the first to describe bacteria and other microbes that he called animalcules (for "tiny animal"). Observing animal, plant, fossil, and mineral specimens, Van Leeuwenhoek illustrated to the world, through his writings, that the ordinary world was and is full of tiny living organisms.
There are many more microbes on the planet than people. In fact, there are more microbes in and on your body than people on Earth: trillions of them, actually. Research continues to reveal the ways in which we live intimately with microbes. Our bodies and environment depend on a healthy diversity of microorganisms to stay healthy.
But not all microbes are beneficial.When a microorganism causes disease or illness, we call it a germ or a pathogen.
Before the discovery of antibiotics and vaccines, infection and illness routinely killed young and old. Throughout history, infectious bacterial and viral diseases have swept through societies and populations. Smallpox, cholera, and the plague are just a few of the viral and bacterial diseases that can now be controlled through vaccination or antibiotics.
Edward Jenner legitimatized the use of vaccination against the variola virus, or smallpox, in 1796. The last natural outbreak of smallpox occurred in the US in 1949. Research with smallpox continues, in the event smallpox is weaponized through bioterrorism.
Before the introduction of penicillin by Alexander Fleming in 1943, bacteria of many kinds invaded human hosts, sometimes killing quickly, sometimes slowly. Penicillin, the first "antibiotic," ushered in an era of medical research that led to the development of many more lifesaving drugs.
The advancement and widespread use of drugs and vaccines saved lives and eliminated suffering, but the overuse of these same substances has led to the serious problem of resistance.

Antimicrobial Resistance — Running Out of Options & Time
For several decades, antimicrobials have been called "wonder" or "miracle" drugs for their ability to deter or eliminate sure death.
As more microbes become resistant to drugs engineered by humans, we are rapidly running out of options to treat serious infections. Front-line drugs (the drugs of choice used to treat infection) are beginning to fail. A couple of examples where we are running out of drug treatment options include:
- Clostridium difficile (CDIFF): Contributing to approximately 15,000 deaths per year, C. difficile bacteria causes potentially fatal diarrhea, usually in elderly, or immune-compromised individuals.
- Carbapenem-resistant Enterobacteriaceae (CRE): Causing an often-fatal infection when it enters the bloodstream, CRE is a family of pathogens (that includes E. coli) that are almost untreatable with medication.
- Candida auris: Resistant to most anti-fungal drugs, Candida auris is a strain of yeast associated with high rates of death among patients where the fungal infection is able to enter the bloodstream.
- Sexually transmitted diseases (STDs): Drugs used to treat increasingly common STDs are rapidly becoming ineffective against syphilis, gonorrhea, and chlamydia infections.
The Most Common Drug-Resistance Terms
This can all be confusing when different terms are used to describe drug resistance. A couple of commonly used terms include:
- Antibiotic resistance: Antibiotic drugs are used primarily against infectious bacteria. You might have gotten a prescription for antibiotics if you had strep throat, caused by Group A streptococcus. Antibiotic resistance is the ability of a bacteria or pathogen to survive in the presence of a particular drug, or drugs. Humans do not become resistant to antibiotics, the bacteria infecting us do. When you take an antibiotic, it should kill the harmful bacteria. Because bacteria reproduce so quickly, some bacteria will genetically mutate and gain the ability to survive the drug. Bacteria can spread resistance to other bacteria. As well, some bacteria are naturally resistant to certain antibiotics, a capability they have carried for millions of years. Because of the widespread use of antibiotics to treat humans and food animals, even when infection was not present, antibiotic resistance has become an urgent public health issue.
- Antimicrobial resistance: Bacteria are microbes. The term "antimicrobial resistance" covers a wider range of microorganisms becoming resistant to drugs, including bacteria, fungi, viruses, and parasites.
- Multi-drug resistant (MDR): When a microbe is resistant to more than one drug, it is called "multi-drug resistant."
- Anti-viral resistant: Viruses are also capable of developing resistance to medications used to treat them.
The Dangers of Resistance
Antimicrobial drugs are used in a wide sweep of industries, including human healthcare, veterinary care, poultry and fish farming, and raising livestock. The issue, and the discussion, of drug-resistant microbes is of concern to those who prescribe, use, or ingest these medications.
On a federal and international level, public health agencies are moving forward with initiatives to reduce unnecessary use of antimicrobial drugs, develop new drugs and new methods to attack infection (including the use of bacteria that eat bacteria!), and provide the general public and medical community with needed information to discuss and proactively respond to this developing health crises.
In 2014, The Review on Antimicrobial Resistance found that unless action is taken now, the number of people dying from resistant infections could reach 10 million by 2050.
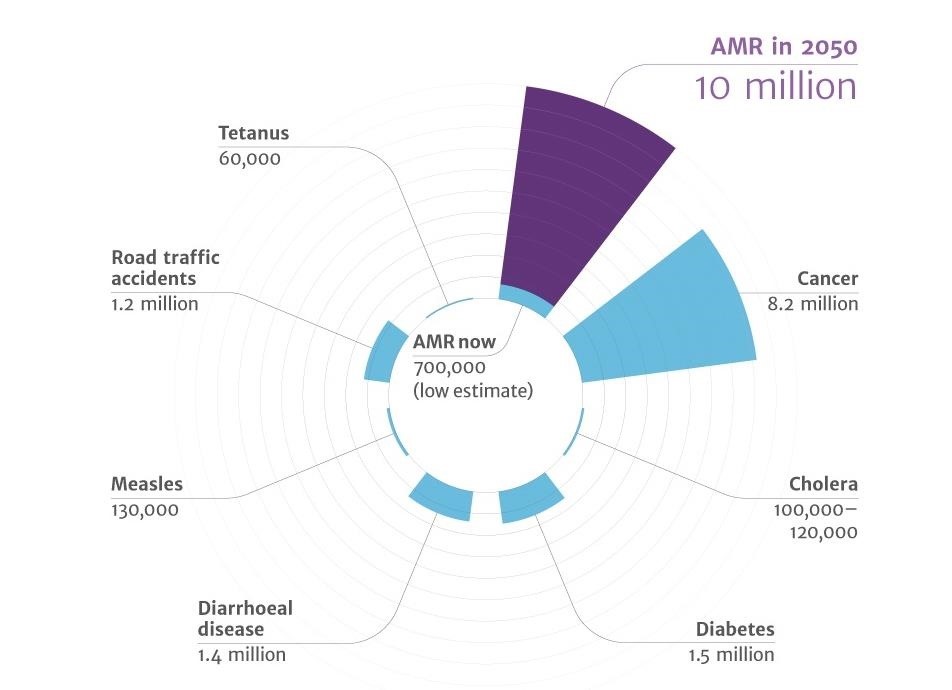
Sifting through family stories, most people have an old recollection of a family member who died of appendicitis, septic shock, or some other infection. Maryn McKenna was moved to respond to the antibiotic crises by the story of her great-uncle who died too soon from an infection that was deadly then — and may soon be deadly again.
The issue becomes starkly personal when you, or a loved one, is diagnosed with a resistant infection. We live in a world, and in bodies, infused with beneficial, neutral, and harmful microbes. A healthy future lies in a discussion of supporting cutting-edge research, developing surveillance infrastructure, addressing socioeconomic conditions, and understanding how we can better use the drugs we do have at present.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.




















Be the First to Comment
Share Your Thoughts